Last Updated: 7:14 p.m.
As confirmed Flagler County Covid-19 cases reach 28 Friday evening and Florida’s exceed 10,000, jumping by more than 1,200 in the last 24 hours, with 170 deaths, local health officials are now stopping short of using reassuring language, saying the worst is yet to come, while various shifts point to the increasing severity of the coronavirus pandemic even locally.
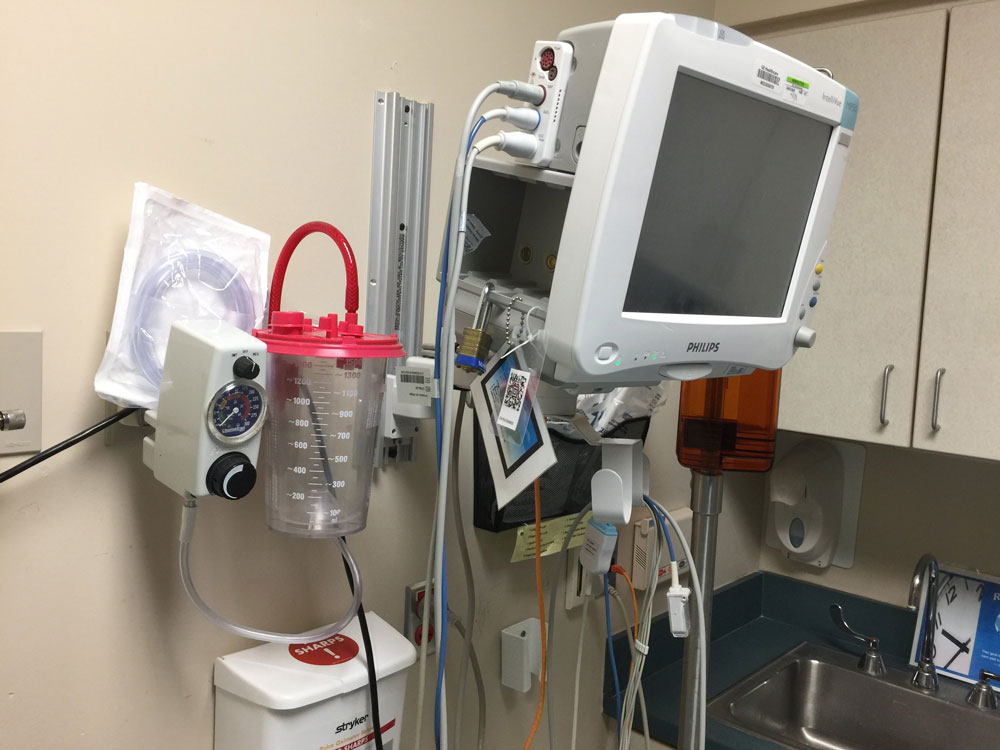
Nevertheless they stress that for now Flagler County’s hospital–AdventHealth Palm Coast–is “in good shape,” with 15 ventilators, while readying to expand ICU capacity threefold in case of a surge, from 18 rooms to 66. The hospital is also exploring using anesthesia equipment to supplement its ventilators.
In yet another escalation of apprehension over the extent of infections, local department of health officials now echo the Centers for Disease Control’s new directive that wearing masks in public is recommended. In another shift, local officials also now acknowledge that more detailed information about where people are infected, down to zip codes and neighborhoods, is necessary, but not yet available.
Testing remains woefully low in the county, with just 290 tests conducted as of this morning, according to Department of Health figures, suggesting that Flagler County’s true extent of the contagion is far higher than the figures posted by the department. But rapid tests promised with much fanfare since last week by President Trump, Gov. Ron DeSantios and even Bob Snyder, chief of the Flagler County Health Department, are not arriving any time soon. Federal officials had promised that they would start arriving at the end of this week. That’s not happening.
The hospital’s ICU plans were detailed by Chief Operating Officer Wally de Aquino in his latest appearance on WNZF’s daily morning update on the coronavirus emergency.
“Regarding capacity, We have certain wings of our hospitals [where] we can make necessary changes to make those rooms identical to an ICU room, de Aquino said. “So the hospital has 18 officially licensed ICU rooms. Well, that would not help us if we get a surge, right? So we identified at least three other wings, each wing has 16 rooms, that we can actually overflow and use as an ICU, which would give us plenty of ICU-like rooms, and I’m using the word ‘like’ because obviously they’re not licensed. But we will be allowed to use them. Some of them we’ll be making some minor modifications and we’ll be looking into that, but thankfully some of those wings already had the proper hookups behind the walls so we can utilize it as a proper ICU room. If we don’t get a surge here, we most likely will be able to help other areas where they don’t have enough beds.”
Today Advent Health’s bed capacity was at 33 percent, according to the state Agency for Health Care Administration.
DeAquino said the hospital currently has just 15 ventilators. He would not specify how many are in use (he said he would do so in subsequent days), but said “we’re not even close to using half” of them. “But there are other ways for us to also work with our anesthesia equipment to make them ventilators as well,” de Aquino said, “so we’re looking into that to see how many we can actually change them from being an anesthesia equipment to a ventilator.” The hospital is also in touch with the Advent command center to seek additional help, if it becomes necessary.
Asked if single ventilators have been adapted to be used for two patients, de Aquino said the Palm Coast hospital isn’t doing that, but other hospitals in the Advent system are testing the approach, which has been used successfully in New York.
According to Florida Department of Health data, four Flagler residents are hospitalized for Covid-19. It’s not clear if that figure includes the Flagler Beach resident who died on Wednesday, and who was hospitalized in Daytona Beach. Because she was a Flagler Beach resident, she was counted as such, even though she was hospitalized elsewhere. There have been numerous, ongoing problems with clarity of data from the health department’s state dashboard.
De Aquino spoke very cautiously about being too optimistic despite the county’s relative fortunes so far.
“Where we need to be cautious is, when we talk about a surge, what is that? What does that mean?” he said. “So, we are in good shape right now, we are in good shape to expect, if we have a surge, if numbers double, two, three times, we’ll be fine here. What does the surge mean–that’s the question to answer: we don’t know.” He said the hospital can be helped to remain in good condition if residents respect social distancing rules and limit infections.
“Obviously we haven’t seen the surge that we’re expecting yet, but overall the employees are doing well and they are actually very responsive to all the changes that we have to do,” de Aquino said. In an acknowledgment of the strains the Covid-19 emergency is placing on family relations, especially when patient visits are either severely restricted or disallowed, the hospital is working with longer-term patients to have virtual visitations, providing devices to them if they don’t have any, thus essentially diminishing the effects of the in-person visitation restrictions. Physicians are also consulting with patients who are Covid-19 positive, or who are under investigation for Covid-19, through telehealth.
Meanwhile, the incompleteness of data, and not even knowing the true extent of the contagion locally, makes any forecasting of a downturn in the grimmest numbers very difficult.
“In terms of the war we’re fighting,” said Steven Bickel, a physician and the medical director for the Flagler and Volusia health departments, “the key component that every country has focused on that we’re going to need to focus on is, when can you start bending the curve. That’s where all the action is. If it’s still a steep, upward slope, and it’s near this exponential spread, you’re in trouble. When you can start diminishing the spread, even if it’s increasing but increasing at a lower rate, then you’re starting to make headway.”
Those are the numbers that dictate what strategies that need to be in place, and whether the strategies need to be intensified. “It’s kind of a shifting background there,” Bickel said, “because you’re looking at spread in the community, and then the measures you do change the rate of spread and you’re trying to extrapolate from that what that means for the population, and the data are not clear. There’s a range for all this stuff. It’s pretty tricky stuff, that’s why people will do these models and then plug in different numbers.”
Until now, and under command from the state department of health, local officials have been resistant to detailing any information about the whereabouts of people with Covid-19 or people under investigation. That may change.
“We would really benefit from knowing the number of positive cases by zip code and by neighborhood,” Snyder, who’s had long and frequent conversations with local reporters about the data, said. “I’m going to do my best as health officer here to push my colleagues in Tallahassee to please consider this. I think something like this would be very important. However, like other health and medical professionals, we just cannot share detailed information about patients, positive cases, persons under investigation, not only because of HIPAA,” he said of the federal health-record privacy law, but doing so would violate Florida law. The department doesn’t comment on current or past epidemiological investigations, he said, “and the medical community regards that information as sacrosanct.”
Still, he said, zip code and neighborhood data tied to Covid-19 “will give us an indication of the community spread, because so many of the people in our community, they do not have symptoms, yet they could be carrying the virus. So this is why this is so important. I’m on it, and I’ll do my best as one person to get things changed as best I can.”
Shortages continue, with both personal protection equipment and tests. While there are a half dozen places where people can get tested, among them the Health Department, AdventHealth Palm Coast and two urgent care facilities, the Health Department is still at the same 100-test-kit capacity as it was last week, and does not expect to have more capacity any time soon, Snyder said. “It’s sufficient for now, we have ordered more, we expect delivery soon,” Snyder said. But he cannot tell when.
Attention had focused on a company’s pledge late last week that it would provide 50,000 rapid tests a day, with results from the tests available in minutes.
“That’s a whole new ballgame,” Trump said in a Rose Garden news conference this week. “I want to thank Abbott Labs for the incredible work they’ve done. They’ve been working around-the-clock.”
Snyder, too, was essentially deceived.
“We are very, very excited about hearing about Abbot labs who are producing 50,000 test kits a day,” Snyder said as recently as Monday, “and it takes 15 minutes for the test results to surface. We would be interested as soon as possible to start some kind of more broad-based surveillance testing. We look forward to that day.”
Yet a document circulated among officials at the Department of Health and Human Services and the Federal Emergency Management Agency this week shows that state and local public health labs were set to receive a total of only 5,500 coronavirus tests from the giant manufacturer of medical devices, diagnostics and drugs, according to emails obtained by Kaiser Health News.
That number falls well short of the “about 500,000 capacity of Abbott tests that” Dr. Deborah Birx, the White House coronavirus response coordinator, said were in the states and were “not being utilized.” Although it is unclear whether she was referring to just the quick test or combining it with another test — one with slower results — that Abbott previously received authorization to sell.
Millions of tests are urgently needed as the virus keeps communities across the country in lockdown and hospitals are overwhelmed with patients.
Labs in all 50 states were set to receive roughly the same number of Abbott’s test cartridges and the devices on which they run ― 100 tests and 10 or 15 devices — the document shows, regardless of how many confirmed COVID-19 cases officials had reported in each state.\
Abbott Labs spokesperson Darcy Ross on Thursday said the company had shipped tests to customers in 18 states but did not elaborate on how many were public clients or governments as opposed to private health care facilities. Ross also said the document circulated among federal officials showed an “intended purchase by HHS and FEMA” of tests and related instruments.
The Federal Emergency Management Administration referred questions to HHS, whose spokesperson Mia Heck said, “We do not comment on any allegedly leaked documents.”
“We can confirm that the federal government is looking to make the initial purchase of a rapid point-of-care test to increase COVID-19 testing capacity in the United States,” she said. “Initially, each state will receive 15 point-of-care instruments, and then they will be able to resupply through the commercial market.”
Accuracy has been a broad problem in the testing for the novel coronavirus. Abbott Labs declined to address specifics on the clinical accuracy of its tests, which was fast-tracked through the Food and Drug Administration’s review process, saying that accuracy data and other performance characteristics will continue to be collected in the field.
State officials are scrambling to obtain Abbott’s highly touted machines after it said they could detect the novel coronavirus in as little as five minutes, or give a negative test result in about 13 minutes. That compares with at least 45 minutes to several days to get results from most of the other types of COVID-19 tests being used.
In announcing the test March 27, Abbott said it was “ramping up production to deliver 50,000 ID NOW COVID-19 tests per day” starting this week to the U.S. health care system.
The price of Abbott’s stock has jumped 26.5% since March 23.
The document circulated among HHS and FEMA officials March 30 and obtained by Kaiser Health News said 5,500 cartridges ― which translates to 5,500 tests — and 780 devices would be shipped to 55 state and local public health labs all over the U.S. An additional 1,200 tests would go to the Pacific territories. Smaller numbers would be sent to the CDC’s lab in Atlanta and the HHS’ Strategic National Stockpile. Abbott has said the company is “working with the administration to deploy the tests to areas where they can have the greatest impact.”
But contrary to the document’s contents, distribution has been far from even. For example, within days of Abbott’s announcing it would ship its rapid test around the country, Detroit Mayor Mike Duggan said he had secured a commitment for five machines and 5,000 tests from the company, making it among the first cities to receive them. The city planned to immediately use them to test police officers and other first responders. A spokesperson for the city didn’t respond to questions Thursday about whether Detroit had the tests in hand.
DeSantis last Saturday said he’d secured just 2,000 such tests. “I think this is really really significant because the problem with the testing we have now is these private labs are overrun. Ideally, they would want to turn it around in 24 to 48 hours but in fact, a lot of people have to wait three, four or five days to be able to get test results,” he said. “I think it also gives a lot of people peace of mind because they at least know what the result is.”
Yet providers have reported rampant problems with COVID-19 tests giving false negatives, in which the virus is not detected even in an infected person. Few medical tests are 100% accurate. Any test can have false negatives or false positives. The problem isn’t unique to COVID-19 tests.
“False-negative test results — tests that indicate you are not infected, when you are — seem to be uncomfortably common,” Dr. Harlan Krumholz, director of the Yale New Haven Hospital Center for Outcomes Research and Evaluation, wrote in a New York Times piece April 1. “Increasingly, and disturbingly, I hear a growing number of anecdotal stories from my fellow doctors of patients testing negative for coronavirus and then testing positive — or people who are almost certainly infected who are testing negative.”
Reasons for this can vary. The test itself might not be so good at picking out true cases from false ones. Or maybe it wasn’t the test at all, but how the sample was taken or how long it had been since the patient began showing symptoms. Doctors and patients should always consider that a result might not be accurate.
That may be especially true now because the COVID-19 test kits from manufacturers and clinical labs became available under emergency use rules.
Under those rules, manufacturers and labs have to submit documentation to the FDA but don’t have to provide as much information as they would under a regular approval process.
Part of the requirement is they show the FDA just how much ― or little — of the virus must exist in the samples before their test can detect it, said Joeffrey Chahine, technical director for the molecular pathology division at MedStar Georgetown University Hospital in Washington, D.C. They also must show how well the tests specifically pick out the coronavirus, not confusing it with another virus, he said.
By relaxing some of the other information normally required, such as correlating results with the status of actual patients, the test kits could get onto the market sooner. And that’s the trade-off.
“This is the downside to loosening the FDA restrictions in that the tests weren’t required to go through the same level of quality assurance, the testing of the test, that we would normally see before they go on the market,” said Marcus Plescia, chief medical officer at the Association of State and Territorial Health Officials. “But I don’t think that was a bad decision. This is an emergency.”
–FlaglerLive and Kaiser Health News



























Extremely Concerned in Flagler County says
Flagler County and Advent Health are so severely unprepared for what is about to transpire that I can’t even bare to think about it. Shame on Flagler County and Advent Health. Shame
Pissed in PC says
Once again the citizens have been lied to by DeSantis and Trump! All these different counties Palm Beach, Orange, etc are only testing about 250 a day before the tests run out. But remember Trump said I like my numbers low when all those people were on the cruise docked in the Pacific Ocean. But please republicans of Flagler keep drinking that kool aid of blissful ignorance.
https://youtu.be/s2QLxGAlu18
Gawd Bless Amerca says
The US was woefully unprepared for the flu of 1918. Woefully unprepared for the great depression of 1929. Woefully unprepared for Pearl Harbor, 9/11, the great recession, and now coronavirus.
But rest assured, we’ll wade through a few miserable years and then go right back to patting ourselves on our back, grovel at the feet of billionaires and unqualified politicians, try to convince the world of how great we are, hug our flag and jerk off with our guns, and then unprepare ourselves for the next avoidable disaster.
Rob Jr says
Anyone who wants a test can get a test.
FlaglerLive, you mean those tests?
They will arrive about the same time the that better, cheaper health care and lower prescription prices shows up.
beachcomberT says
Thanks for a detailed report that zeroes in on the key issues and unknowns. Another aspect which seems to be getting little discussion — what about the mild cases where people are recovering at home? Because of shortage of testing, we don’t know how many that might be. Presumably, no one is getting tested if they feel perfectly fine. If they feel slightly off, but test negative, what happens? They’re sent back home — but are they being told to self-quarantine just to be on the safe side? How about people who test positive but can still breathe OK. Are they being sent home or admitted to a hospital? So far, the stats seems to be showing only the serious cases — those requiring hospitalization because of acute symptoms. We can only guess how many mild cases are recovering in private homes.
Appreciative says
Thank you, FlaglerLive. You are the best source of information we have.
Shark says
Nothing to worry about. T-rump said it will go away when it gets warm !!!!
Sherry says
The Federal Government is failing us completely, and costing lives!
This from the Associated Press:
President Donald Trump’s insistence that the stockpile is only a short-term backup for states, not a commitment to ensure supplies get quickly to those who need them most during an emergency, the latest front in a concerted White House effort to try to put the onus for battling the crisis on the states, with Washington meant to play more of a supporting role.
Trump angrily defended the approach in his Friday news conference, his early sunny demeanor darkening as he was pressed on expected death rates and his son-in-law Jared Kushner’s comments suggesting the national stockpile is not intended for states. He sparred with reporters and insisted his administration was “doing our best for New York,” the pandemic’s epicenter, even as Governor Andrew Cuomo warns the state is in danger of not having enough ventilators to help patients stricken with coronavirus in a matter of days.
The alteration of the language describing the stockpile was reflected on government websites on Friday, a day after Kushner, a White House senior adviser who has taken a larger role in the coronavirus response, offered a new argument about the supplies.
“I leave it up to the governors,” Trump said Friday about whether every state should have a stay-at-home order. “I like that from the standpoint of governing.”
But in statehouses across the nation, there has been increasing frustration at the lack of assistance from Washington.
Trump scoffed at New York Gov. Andrew Cuomo’s request for 30,000 ventilators for the state, a virus hotbed. In neighboring Connecticut, Gov. Ned Lamont told CNN on Tuesday that the state couldn’t get personal protective equipment from the national stockpile and was only able to get 50 ventilators from the federal supply. He said he was told that he’ll have to secure the rest on his own.
States have also reported multiple issues with the equipment the federal government has managed to send.
Illinois was supposed to receive a federal shipment of 300,000 N95 masks in March, but instead received less-effective surgical masks, Gov. J.B. Pritzker revealed during a Tuesday briefing. California Governor Gavin Newsom announced on Twitter last week that he had received 170 broken ventilators from the national stockpile, far less than the 10,000 breathing machines he’s seeking.
Nearly 6,000 medical masks sent to Alabama had dry rot and a 2010 expiration date. More than 150 ventilators sent to Los Angeles were broken and had to be repaired. In Oregon, it was masks with faulty elastic that could cause the straps to snap, exposing medical workers to the disease.
Carolyn says
As of today: Advent is still following cdc guidelines for testing.. asking have you been on a cruise ship or travelled internationally in the past 14 days ? Well, no one has .. so basically that equals =no one qualifies for a test. This county needs testing !
Cynthia says
If there were more tests available, the numbers of positive COVID19 people would increase. Without the tests, more may die unnecessarily, like Mrs. Strickland.
tulip says
The difference between the other catastrophies you mentioned and the coronavirus is that way back when there was no way of knowing that danger was on the way,so they couldn’t really prepare. With the virus, Trump and other countries knew it back at end of Dec,, were warned of it by intellingence and Trump decided it wasn’t worth being concerned about while other countries began stocking up with supplies sent by 3M company.
Had Trump been concerned about the country rather than his ratings and how well he was getting along with Putin and Kim Jong un, we might be a lot better off than we are. It’s disgusting to think we can’t do better than a third world country.
Ld says
Employees are afraid to complain about being directed to REUSE masks, put in a bag for reuse day after day…employees are risking their own health and that of others by adhering to this but can’t afford to lose their jobs. For what the hospital charges patients and insurers they can provide PPE to employees.
tulip says
Well, if Flagler can’t get the testing equipment, that means not many can be tested and it looks like Flagler County has a low count. That makes Trump very happy.
Sm says
How do you prepare for such events??
Candace says
Your leaders have failed you. They chose economy over health. Even now as they pretend to put a stay at home oreder, they allow many non essential buissnesses to stay open including CHURCHES and GUN SALES. Plus many other non essential. Its obvious your states goverment wants to have its cake and eat it too. You are all being placed in harms way.
Random says
Don’t your worry, Biden’s on the job, assuring Trump another 4 years. Dems are paid to lose.
maggie says
Loving this concerted effort by all Florida press today, to assure The People that our medical facilities are “prepared”. That way, you can just relax and go back to binge-watching Netflix, and you won’t have time to think about all the ways that “profiteering” off America’s sick and dying by Shareholders (whose identities are protected) has led us to this LACK of preparedness.
Make no mistake, our local hospitals are not prepared to be overrun with thousands of sick people. One look at New York City should make that crystal clear to all of us.
Don’t let all the soothing psych-outs fool you, my friends and neighbors. Keep talking about New York. Keep talking about how our For Profit “Health Care” system needs to be scrapped, because no one should ever profit off sick or dying people. It’s starting to scare the upper-crust…
Concerned Citizen says
Somehow I’m not surprised that the test kits haven’t shown. Just like I’m not surprised that Advent Health wasn’t prepared for a Pandemic.
I’m also not surprised that non of our Leadership have stepped forward to handle this rapidly declining situation. There have been no “fire side” speeches reassuring citizens. Hell where is our leadership anyways?
A major event hit our area and is an ongoing crisis. Yet non of our politicians have stepped forward to say let’s fix this. And now. I have been imoressed with Jonathan Lord and his management on the Emergency Management side. But where is his support?
We deserve better than this yet all we have is a bunch of old corrupt men that have dissapeared when the going got tough and the money started drying up.
I will remember this if and when we are allowed to vote this year. And you should to.
Pogo says
@The citizens and economy of our country are smothering to death
The jabbering fat criminal a minority of all voters put in charge is having a infantile tantrum/total breakdown in front of the whole world.
Everyone – EVERYONE – needs to get up off their ass and demand trump resign immediately.
Speak now as if your very life depends on it – it does.
https://www.google.com/search?&q=contact+the+president
Steve says
News flash, Pathological liars lie and what was expressed yesterday has no influence on the lie or take they want to convey here in the present. Tomorrow is just another blank canvas to misinform, misguide and appease those blind followers who dont have their own form of self Independence, determination and dont or cant face the reality of factually opposing views while taking it all personal rather than differing of opinions. My take is this will end badly, worse than it needed to be with proper Leadership in place.
Greg Matthews says
Great job being done by Advent Health. Thank goodness we have a hospital system that is ahead of the curve and has a vast network to reach out to.
And thank you to all on the front line if this war!
Neil Nelson says
Ventilators are of no use if you do not have the injectable medications that go with them, and guess what…….there is now a nationwide shortage developing of these injectable meds…..as Covd-19 hotspots consume the available supplies. There are few manufacturers of these injectable and a mandatory 21 day sterility cycle built into their Manufacturing Cycle.
Three are several different injectablees required (Fentamyl among them and also paralytics) and the absence of any one limits the ability of the Physician to effectively use the ventilator to treat a patient
Sherry says
Critical, “LIFE and DEATH” questions for which there seems to be little response, except to BLAME those asking the questions. WHAT EXACTLY IS THE PLAN FOR RECOVERY ?
https://www.cnn.com/videos/politics/2020/04/05/jake-tapper-trump-coronavirus-covid-19-plan-sotu-vpx.cnn
Sad Times says
Sadly, most of these problems are due to Republicans…who only care about money and making money…no matter how. The Rs screw up and the Ds try their best to fix things.
I just wish that the Rs cared about “we, the people.” But they don’t. Just look at the daily briefings…talk of death and approaching death…not any attempts at emotion or a tear from our President or Vice President. I keep hoping, though.
John Staub says
Our Governor and President are being lied too. This should have killed thousands by now. According to the experts have given us info on from the beginning. I’m still waiting quite honestly. I understand no one wants to take a chance. Statements made from experts abroad have told us we will see mass deaths (when?) Next year I guess.
H1N1 Swine flu is 1 of 2 flu strains currently floating around. But yet, I bet no one knows this. (ON CDC look it up) Due to all the issues and frankly speaking frenzy of warding off the Covid-19, we have kept that in check. actually a lot of sickness is squelched I presume. Don’t know for sure. But my guess is this will continue as is till November 4, 2020. And if President Trump wins. It will continue. But no worries we shall have access to the new vaccine for this horrible virus that is killing hundreds of thousands of Americans.
Free your mind turn off the tv. Go outside get Vitamin D, Exercise and live life.
And the elite are getting what they want… division of the masses causing fear and panic. We didn’t act like this in 2009 (Swine Flu) and yet thousands and thousands died. And yet was treated lightly. Why now? The elite (Not Trump) Are creating this nonsense. Combat Covid-19 the right way. Not but causing panic and division.
Our people and the small businesses are suffering too. Don’t worry the rich don’t care it does not effect them much at all. from the poorest to the middle class get devastated on the fear of what”MIGHT” happen. How come we speed on all the roadways that can kill too. And yet we chance it because we are in a hurry right? Keep in mind we need more coming together than dividing.
But because everyone is on edge, understandably. No need to not use our heads.
Live life to the fullest! We live where everyone else vacations, PARADISE!
Karen says
110,00 residents in Flagler County and 15 ventilators? Hmmmmmm
Carry says
If you feel unsafe at your job you can quit and collect unemployment under the new stimulous guidlines.
Agkistrodon says
They are also allowing abortions as that is a constultitutional right it seems, should they stop those too?
Agkistrodon says
Why didn’t Obama stock pile ventilators and m95s while he was in office, he had 8 years to prepare, seemshedidnt either, after all a whole lot if people died from swine flu……